Publications
Below are some selected scientific publications we have enabled 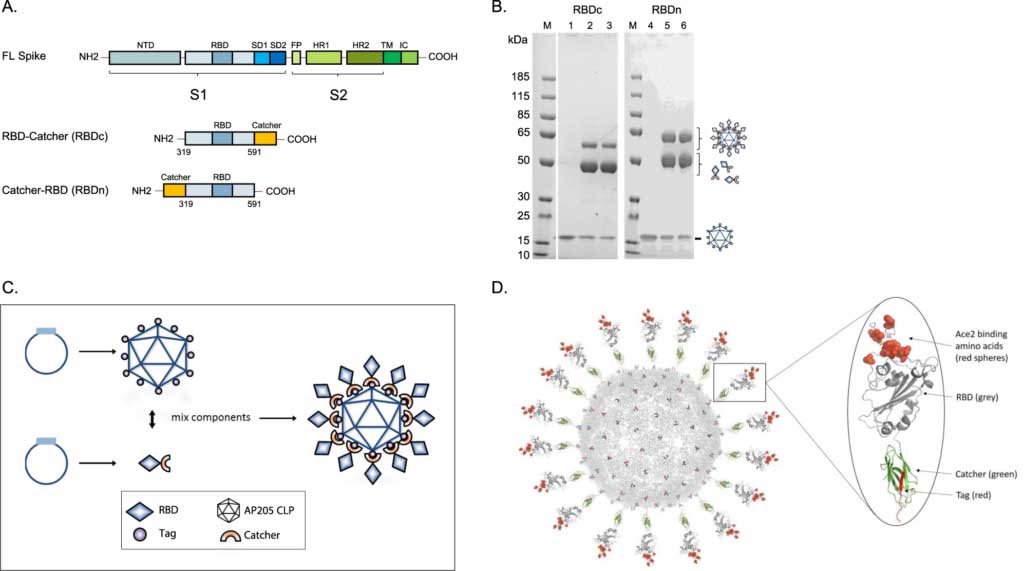 .
.
For a list of all 150+ publications click here
Capsid-like particles decorated with the SARS-CoV-2 receptor-binding domain elicit strong virus neutralization activity
The rapid development of a SARS-CoV-2 vaccine is a global priority. Here, we develop two capsid-like particle (CLP)-based vaccines displaying the receptor-binding domain (RBD) of the SARS-CoV-2 spike protein. RBD antigens are displayed on AP205 CLPs through a split-protein Tag/Catcher, ensuring unidirectional and high-density display of RBD. Both soluble recombinant RBD and RBD displayed on CLPs bind the ACE2 receptor with nanomolar affinity. Mice are vaccinated with soluble RBD or CLP-displayed RBD, formulated in Squalene-Water-Emulsion. The RBD-CLP vaccines induce higher levels of serum anti-spike antibodies than the soluble RBD vaccines. Remarkably, one injection with our lead RBD-CLP vaccine in mice elicits virus neutralization antibody titers comparable to those found in patients that had recovered from COVID-19. Following booster vaccinations, the virus neutralization titers exceed those measured after natural infection, at serum dilutions above 1:10,000. Thus, the RBD-CLP vaccine is a highly promising candidate for preventing COVID-19.
Nature Communications volume 12, Article number: 324 (2021).
Cyrielle Fougeroux, Louise Goksøyr, Manja Idorn, Vladislav Soroka, Sebenzile K. Myeni, Robert Dagil, Christoph M. Janitzek, Max Søgaard, Kara-Lee Aves, Emma W. Horsted, Sayit Mahmut Erdoğan, Tobias Gustavsson, Jerzy Dorosz, Stine Clemmensen, Laurits Fredsgaard, Susan Thrane, Elena E. Vidal-Calvo, Paul Khalifé, Thomas M. Hulen, Swati Choudhary, Michael Theisen, Susheel K. Singh, Asier Garcia-Senosiain, Linda Van Oosten, Gorben Pijlman, Bettina Hierzberger, Tanja Domeyer, Blanka W. Nalewajek, Anette Strøbæk, Magdalena Skrzypczak, Laura F. Andersson, Søren Buus, Anette Stryhn Buus, Jan Pravsgaard Christensen, Tim J. Dalebout, Kasper Iversen, Lene H. Harritshøj, Benjamin Mordmüller, Henrik Ullum, Line S. Reinert, Willem Adriaan de Jongh, Marjolein Kikkert, Søren R. Paludan, Thor G. Theander, Morten A. Nielsen, Ali Salanti & Adam F. Sander
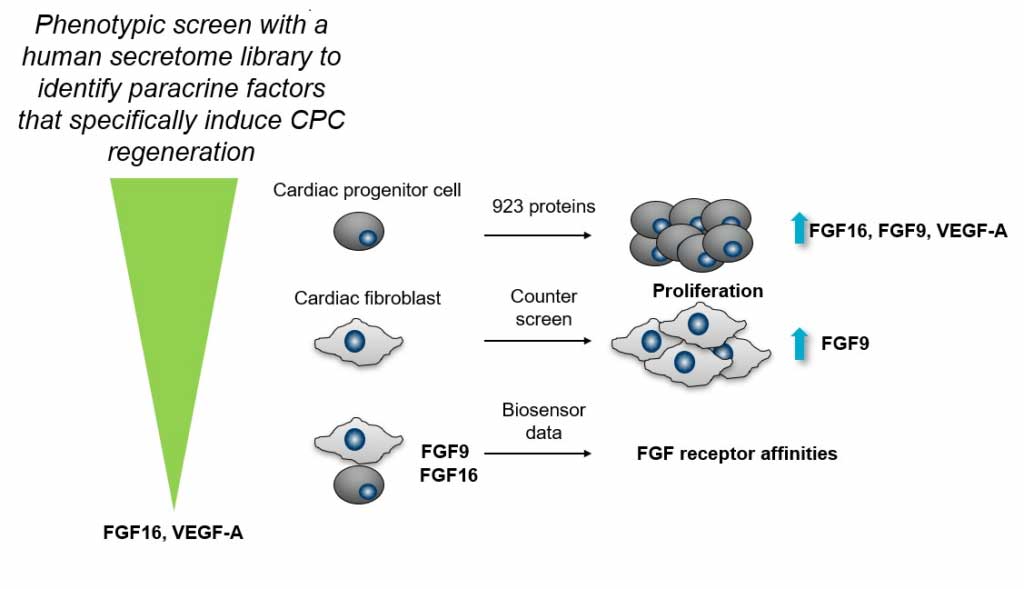
Phenotypic Screen with the Human Secretome Identifies FGF16 as Inducing Proliferation of iPSC-Derived Cardiac Progenitor Cells
Paracrine factors can induce cardiac regeneration and repair post myocardial infarction by stimulating proliferation of cardiac cells and inducing the anti-fibrotic, antiapoptotic, and immunomodulatory effects of angiogenesis. Here, we screened a human secretome library, consisting of 923 growth factors, cytokines, and proteins with unknown function, in a phenotypic screen with human cardiac progenitor cells. The primary readout in the screen was proliferation measured by nuclear count. From this screen, we identified FGF1, FGF4, FGF9, FGF16, FGF18, and seven additional proteins that induce proliferation of cardiac progenitor cells. FGF9 and FGF16 belong to the same FGF subfamily, share high sequence identity, and are described to have similar receptor preferences. Interestingly, FGF16 was shown to be specific for proliferation of cardiac progenitor cells, whereas FGF9 also proliferated human cardiac fibroblasts. Biosensor analysis of receptor preferences and quantification of receptor abundances suggested that FGF16 and FGF9 bind to different FGF receptors on the cardiac progenitor cells and cardiac fibroblasts. FGF16 also proliferated naïve cardiac progenitor cells isolated from mouse heart and human cardiomyocytes derived from induced pluripotent cells. Taken together, the data suggest that FGF16 could be a suitable paracrine factor to induce cardiac regeneration and repair.
International Journal of Molecular Sciences, Volume 20, Issue 23, 2019.
Karin Jennbacken, Fredrik Wågberg, Ulla Karlsson, Jerry Eriksson, Lisa Magnusson, Marjorie Chimienti, Piero Ricchiuto, Jenny Bernström, Mei Ding, Douglas Ross-Thriepland, Yafeng Xue, Diluka Peiris, Teodor Aastrup, Hanna Tegel, Sophia Hober, Åsa Sivertsson, Mathias Uhlén, Per-Erik Strömstedt, Rick Davies and Lovisa Holmberg Schiavone
Virus-like particle display of HER2 induces potent anti-cancer responses
Overexpression of human epidermal growth factor receptor-2 (HER2) occurs in 20–30% of invasive breast cancers. Monoclonal antibody therapy is effective in treating HER2-driven mammary carcinomas, but its utility is limited by high costs, side effects and development of resistance. Active vaccination may represent a safer, more effective and cheaper alternative, although the induction of strong and durable autoantibody responses is hampered by immune-tolerogenic mechanisms. Using a novel virus-like particle (VLP) based vaccine platform we show that directional, high-density display of human HER2 on the surface of VLPs, allows induction of therapeutically potent anti-HER2 autoantibody responses. Prophylactic vaccination reduced spontaneous development of mammary carcinomas by 50%-100% in human HER2 transgenic mice and inhibited the growth of HER2-positive tumors implanted in wild-type mice. The HER2-VLP vaccine shows promise as a new cost-effective modality for prevention and treatment of HER2-positive cancer. The VLP platform may represent an effective tool for development of vaccines against other non-communicable diseases.
OncoImmunology, Volume 7, Issue 30, 2018.
Arianna Palladini, Susan Thrane, Christoph M. Janitzek, Jessica Pihl, Stine B. Clemmensen, Willem Adriaan de Jongh, Thomas M. Clausen, Giordano Nicoletti, Lorena Landuzzi, Manuel L. Penichet, Tania Balboni, Marianna L. Ianzano, Veronica Giusti, Thor G. Theander, Morten A. Nielsen, Ali Salanti, Pier-Luigi Lollini, Patrizia Nanni & Adam F. Sander
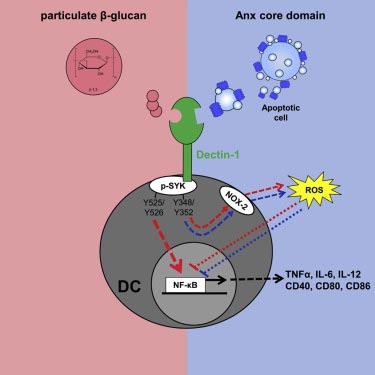
Dectin-1 Binding to Annexins on Apoptotic Cells Induces Peripheral Immune Tolerance via NADPH Oxidase-2
Uptake of apoptotic cells (ACs) by dendritic cells (DCs) and induction of a tolerogenic DC phenotype is an important mechanism for establishing peripheral tolerance to self-antigens. The receptors involved and underlying signaling pathways are not fully understood. Here, we identify Dectin-1 as a crucial tolerogenic receptor binding with nanomolar affinity to the core domain of several annexins (annexin A1, A5, and A13) exposed on ACs. Annexins bind to Dectin-1 on a site distinct from the interaction site of pathogen-derived β-glucans. Subsequent tolerogenic signaling induces selective phosphorylation of spleen tyrosine kinase (SYK), causing activation of NADPH oxidase-2 and moderate production of reactive oxygen species. Thus, mice deficient for Dectin-1 develop autoimmune pathologies (autoantibodies and splenomegaly) and generate stronger immune responses (cytotoxic T cells) against ACs. Our data describe an important immunological checkpoint system and provide a link between immunosuppressive signals of ACs and maintenance of peripheral immune tolerance.
Cell Reports, Volume 29, Issue 13, 2019.
Kevin Bode, Fatmire Bujupi, Corinna Link, Tobias Hein, Stephanie Zimmermann, Diluka Peiris, Vincent Jaquet, Bernd Lepenies, Heiko Weyd, Peter H. Krammer
Inhibition of Tumor Cell Growth and Cancer Stem Cell Expansion by a Bispecific Antibody Targeting EGFR and HER3
The frequent activation of HER3 signaling as a resistance mechanism to EGFR-targeted therapy has motivated the development of combination therapies that block more than one receptor tyrosine kinase. Here, we have developed a novel tetravalent, bispecific single-chain diabody-Fc fusion protein targeting EGFR and HER3 (also known as ErbB3) that integrates the antigen-binding sites of a humanized version of cetuximab as well as a recently developed anti-HER3 antibody, IgG 3-43. This bispecific antibody combines the binding and neutralizing properties of the parental antibodies, as observed in biochemical and in vitro two-dimensional and three-dimensional cell culture assays, and gave rise to long-lasting growth suppression in a subcutaneous xenograft head and neck tumor model. In triple-negative breast cancer (TNBC) cell lines, treatment with the bispecific antibody inhibited the proliferation and oncosphere formation efficiency driven by HER3 signaling. In an orthotopic MDA-MB-468 tumor model, this translated into antitumor effects superior to those obtained by the parental antibodies alone or in combination and was associated with a reduced number of cells with stem-like properties. These findings demonstrate that the bispecific antibody efficiently blocks not only TNBC proliferation, but also the survival and expansion of the cancer stem cell population, holding promise for further preclinical development.
Targeting Human Cancer by a Glycosaminoglycan Binding Malaria Protein.
In a study made by Ali Salanti et al. regarding binding between human cancer cells and glycosaminoglycan binding malaria protein (VAR2CSA). The study utilises Attana QCM technology to perform binding studies using both cell based and biochemical approaches.
Cancer Cell, Volume 28, Issue 4, p. 500–514, 2015.
Ali Salanti, Thomas M. Clausen, Mette Ø. Agerbæk, Nader Al Nakouzi, Madeleine Dahlbäck, Htoo Z. Oo, Sherry Lee, Tobias Gustavsson, Jamie R. Rich, Bradley J. Hedberg, Yang Mao, Line Barington, Marina A. Pereira, Janine LoBello, Makoto Endo, Ladan Fazli, Jo Soden, Chris K. Wang, Adam F. Sander, Robert Dagil, Susan Thrane, Peter J. Holst, Le Meng, Francesco Favero, Glen J. Weiss, Morten A. Nielsen, Jim Freeth, Torsten O. Nielsen, Joseph Zaia, Nhan L. Tran, Jeff Trent, John S. Babcook, Thor G. Theander, Poul H. Sorensen and Mads Daugaard
Real-time and label free determination of ligand binding-kinetics to primary cancer tissue specimens; a novel tool for the assessment of biomarker targeting
The paper describes use of Attana QCM label free methodology to study molecular binding kinetics in situ on FFPE tissues. FFPE tissue sections of human placenta tissues and prostate cancer were immobilised on COP-1 surfaces using ploy-L-lysine solution. Interaction between vAR2 protein and its receptor has been analysed using Attana Cell 200 system. Affinity value in the nanomolor range was detected for the interaction between rVAR2 and its receptor and no detectable binding was observed with the haelthy tissues.
Sensing and Bio-Sensing Research, Volume 9, p. 23-30, 2016.
Thomas Mandel Clausen, Marina Ayres Pereira, Htoo Zarni Oo, Mafalda Resende, Tobias Gustavson, Yang Mao, Nobuo Sugiura, Janet Liew, Ladan Fazli, Thor G Theander, Mads Daugaard and Ali Salanti
Cellular glycosylation affects Herceptin binding and sensitivity of breast cancer cells to doxorubicin and growth factors
This study explores the effect of cell surface glycans on cancer therapy.
Scientific Reports 7, Article: 43006, 2017.
Diluka Peiris, Alexander F. Spector, Hannah Lomax-Browne, Tayebeh Azimi, Bala Ramesh, Marilena Loizidou, Hazel Welch and Miriam V. Dwek
Label-free in-flow detection of receptor recognition motifs on the biomolecular corona of nanoparticles
Proteins and other biomolecules in human biological fluids interact with the surfaces of nanoparticles. These interactions generate a coating (corona) around the nanoparticles which is the nanoparticles interface in the human body. Attana´s technology was used to characterized corona coated nanoparticles interactions with different biomolecules. The obtained information is a valuable tool to optimize nanoparticles for therapeutic applications and to improve the predictability of in vivo performance of the nanoparticles. During the EU-project several different assays have been developed based on Attana’s technology and some of the results have now been published in the journal Nanoscale. In the paper, accessible functional epitopes of transferrin-coated nanoparticles are quantified and their number is correlated to differences in nanoparticle size and functionalization. The label free in flow target recognition pushes the assays into a more in vivo-like scenario than previous technologies. The assays are applicable to a wide array of nanoparticles and consequently hold the potential to become a standard technique for the classification of nanoparticles based on their biological external functionality.
Nanoscale, vol. 10, 2018, p. 5474-5481.
M. Gianneli, E. Polo, H. Lopez, V. Castagnola, T. Aastrup & K.A. Dawson.
Combined Bacteria Microarray and Quartz Crystal Microbalance Approach for Exploring Glycosignatures of Nontypeable Haemophilus influenzae and Recognition by Host Lectins
The paper describes the use of the Attana Cell 200 instrument to characterize the glycosignature of bacteria (haemophilus influeanze). The bacteria have been immobilized using a ConA-LNB surface. Different lectins injected and the binding kinetic analyze. Comparison of the frequency shift obtained using different strains confirm the result obtained in micro-array.
Analytic Chemistry, Volume 88, Issue 11, p. 5950–5957, 2016.
Ioanna Kalograiaki, Begoñ a Euba,Davide Proverbio, María A. Campanero-Rhodes, Teodor Aastrup, Junkal Garmendia and Dolores Solís
Projects & collaborations
NanoCarb: Development of glycosylated nanoparticles with enhanced therapeutic actions
NanoCarb: Development of glycosylated nanoparticles with enhanced therapeutic actions
Participants – Royal College of Surgeons in Ireland, University of Warwick, Università degli Studi di Milano, CIC biomaGUNE, Vlaamse Instelling voor Technologisch Onderzoek, Ludger Ltd, Midatech Pharma, Mario Negri Institute for Pharmacological Research.
The overarching aim of the NanoCarb ETN project is to develop glycosylated nanoparticles with enhanced therapeutic actions for healthcare applications. 15 Early Stage Researchers (ESRs) recruited for the NanoCarb project will work on their individual ESR projects designed to provide appropriate research goals for PhD students. Each ESR will be exposed to a well-balanced and optimal experience in academia, industry and research centre in order to increase their research experience and scientific maturity.

Ultra-sensitive micro-sensors
Participants: Linnaeus University, Luma Metall AB and Attana AB.
The challenges facing Society pertaining to quality of life through health management and food supply, public security and sustainability are driving a demand for more sensitive, more reliable, smaller and cheaper devices for use in clinical, industrial, security and environmental diagnostic contexts. Our consortium shall develop new nanotechnology-based diagnostic strategies for the highly ultra-sensitive detection of substances using either electrochemical-, piezoelectric- or fluorescence-based detection. Through a collaboration of Attana AB, LUMA-Metall and Linnaeus University with expertise in sensor development and commercialisation, ultra-thin metal-based development and nanomaterial design and development, respectively. We shall establish novel chemistries for surface derivatisation and polymeric nanoparticle synthesis that shall be developed using Attana AB’s quartz crystal microbalance technology-platform. These chemistries shall then be used to produce metal-polymer or metal-biopolymer material hybrids using the LUMA-Metall AB’s exclusive ultra-thin wire formats. Initially, the ultra-thin surfaces and micrometer-diameter wire-based materials shall be employed to explore detection of proof-of-concept targets in these sensor platforms. Candidate systems shall then be used to develop sensors selective for import and and difficult-to-measure analytes, in particular:
a) peptide drug determination in plasma
b) cancer biomarkers for earlier diagnosis
c) mycotoxins in food stuffs
An assessment of potential intellectual property development and commercial viability after benchmarking with established methods.
Concluded projects
Development of CD34/CD3 bispecific antibodies for treatment of leukemia and preconditioning before allogeneic hematopoietic stem cell transplantation
Participants – Karolinska Universitetsjukhuset, Kungliga Tekniska Högskolan, Attana AB.
The aim of the project Specific aims of the project to design a range of bispecific antibodies towards CD34 (stem cells and leukemic cells) and CD3 (T cells) and to identify and select lead candidates by evaluating binding properties towards cell-surface bound antigen by Attanas’ cell based assays.
CellSenseGels: Development of sensitive and robust cell-based biosensor assay platforms using peptide gel matrices
Participants – University Stuttgart, Biogelx Ltd, Attana AB.
The project will develop a robust sensitive platform for measurements of molecular kinetics, using peptide hydrogel technology as a 3D matrix. The gels will offer a tissue-mimicking environment, ensuring increased biological relevance and greater data quality. Furthermore, this sensor will improve the range and data quality of biosensor assays through increased signal strength and reduced background noise. Product will be cell-type optimized kits incorporating customised gel-matrices.
NanoClassifier: QCM for rapid label-free Bionano interface evaluation and screening of effectiveness of nano-targeting strategies for therapeutics.
Participants – University College Dublin, Attana AB.
Nanomedicine and Nanosafety rely on the same fundamental interactions between the nanoparticle interface and the biological milieu surrounding it, and it is this “corona” of proteins and other biomolecules that form at the bionanointerface that determines the fate and behaviour of nanomaterials. Understanding, classifying and predicting nanoparticle coronas from their physicochemical properties offers a novel approach to screening for toxicity at early stages of product development and for regulatory purposes.
Quality Control and Purification of New Biological Pharmaceuticals
Participants: Astra Zeneca AB, Attana AB, Akzo Nobel Pulp and Performance Chemicals AB and Karlstad University and three more partners.
Thursday November 30th 2017 the project BIO-QC “Quality Control and Purification of New Biological Pharmaceuticals” was initiated. Biological pharmaceuticals are proteins or similar molecules, which mimics molecules in the body and are injected in the body to stimulate the immune system. To provide criteria’s for quality control of biological pharmaceuticals.
The project has a budget of approximately €2 million and includes seven different parties, Attana AB, AstraZeneca, Akzo Nobel Pulp and Performance Chemicals AB and Ridgeview Instruments AB and Karlstad University, Linné University and Örebro University.